Implement Evidence-Based Medicine for Better Mental Health
Learn everything about nootropics, mental health, and biohacking. It is never too late to stimulate, entertain and inspire yourself.
Featured On




What You Find Here
Our purpose at EvidenceLive is to break the myths around mental health and present you latest evidence-based knowledge on utilizing proper nutrition with lifestyle changes to optimize your brain's potential.

Brain Health
A healthy brain is a key to your overall well-being, which can help you to be stronger physically and mentally.

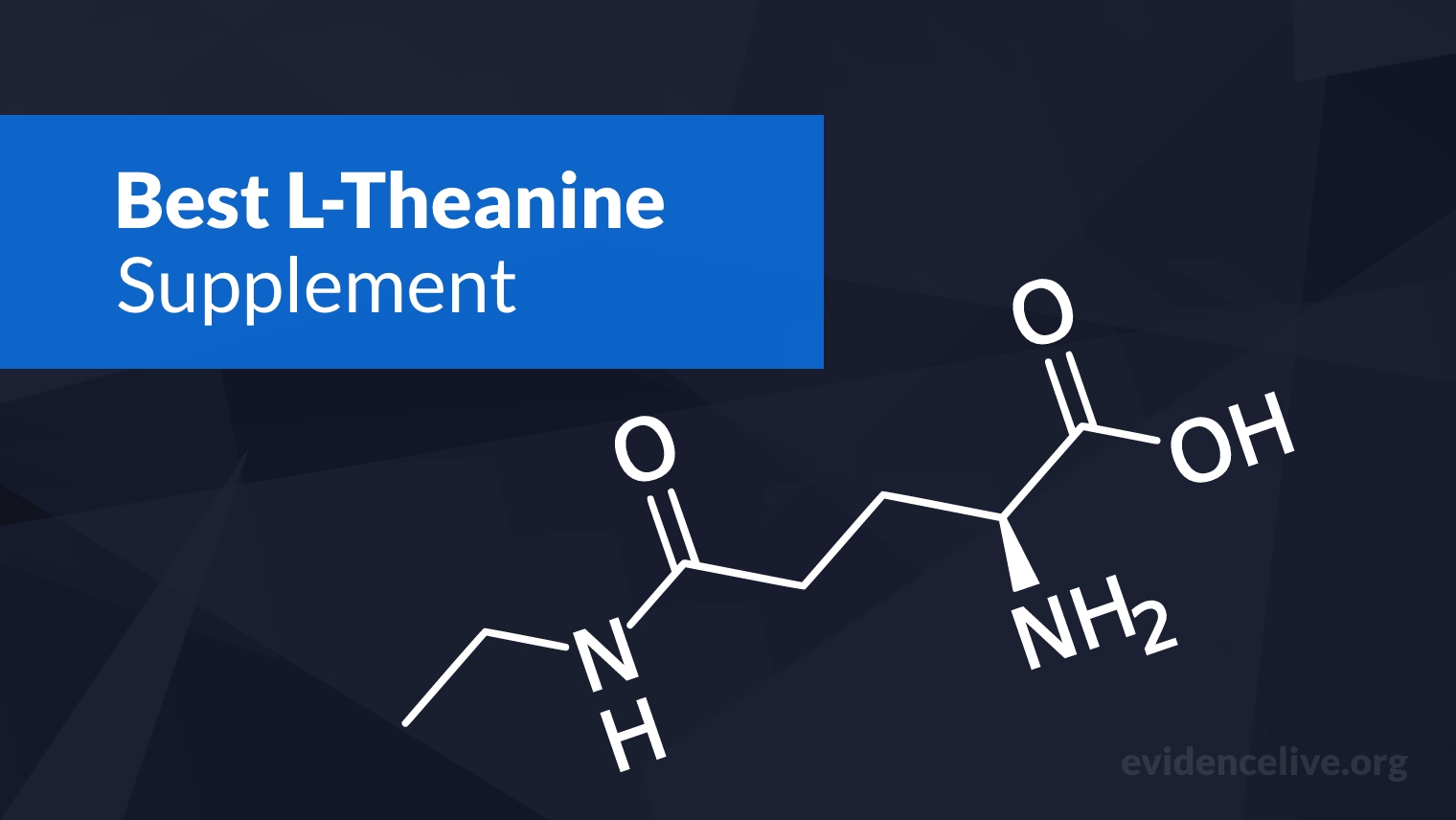
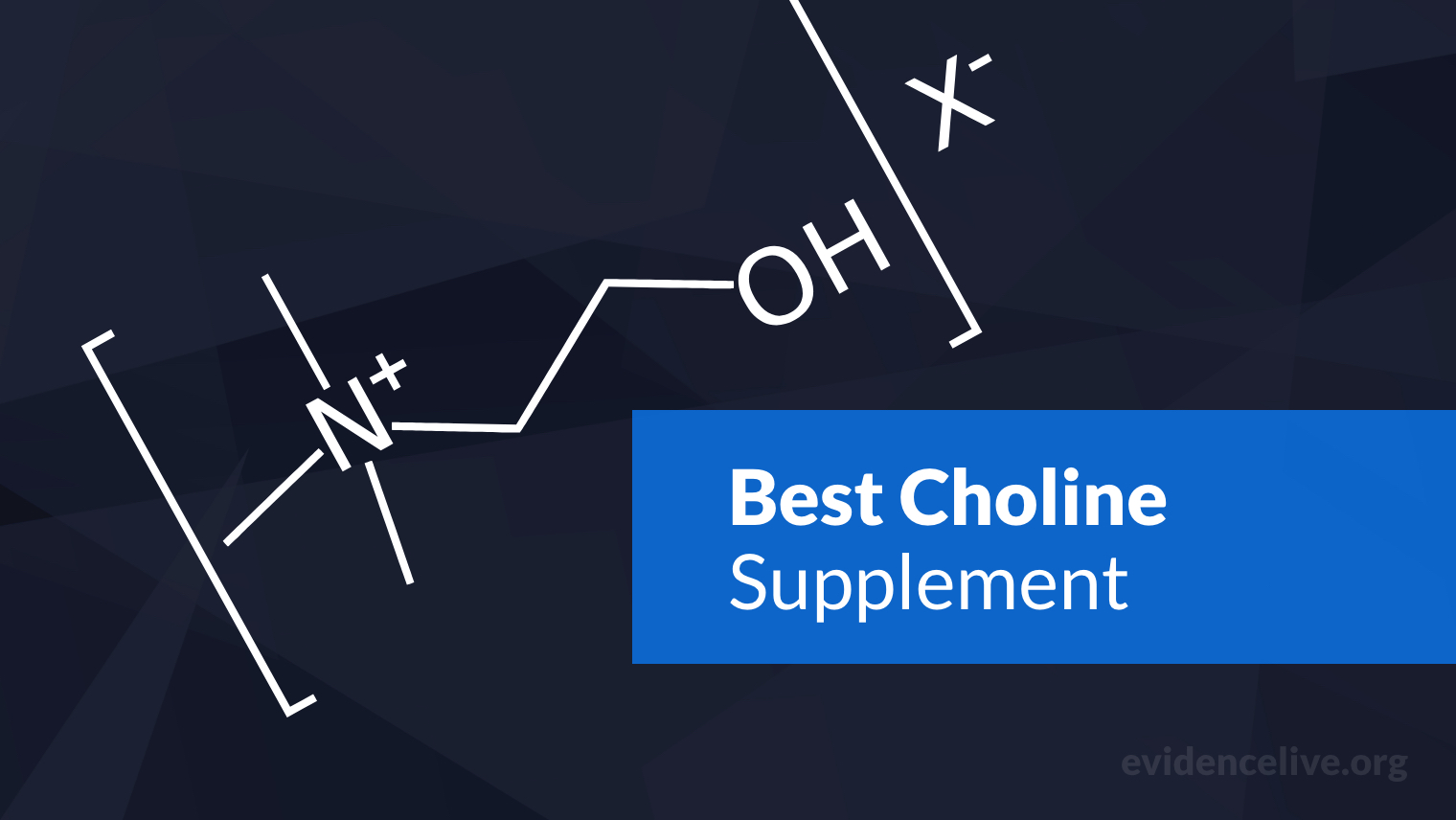
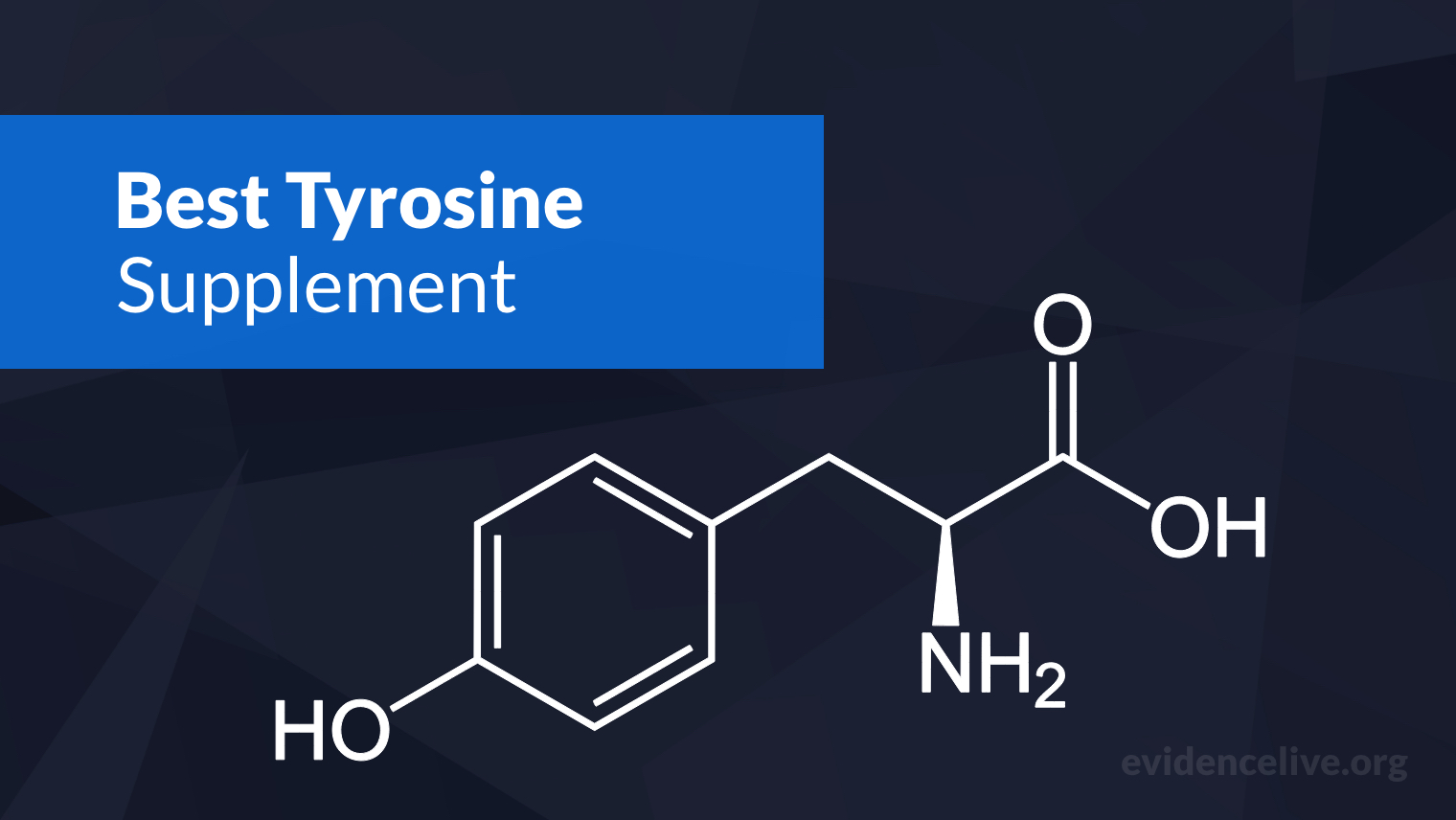
Nootropics
We have compiled all the latest research on nootropics in one place so you can make informed choices.

Reviews
Our trustworthy reviews and in-depth guides will help you to find the right nootropic supplement for your needs.